AI in Healthcare Pulse — 2026-04-24
This week saw mounting scrutiny of federal AI prior authorization programs delaying patient care, a landmark NEJM perspective on autonomous AI managing prescription renewals, and record-breaking Q1 digital health funding driven by AI drug discovery. Nature Medicine weighed in with a pointed editorial questioning whether clinical AI is actually improving care.
AI in Healthcare Pulse — 2026-04-24
Regulatory & Policy Watch
1. Federal AI Prior Authorization Test Delays Care for Seniors
- What happened: A federal Medicare pilot program called WISER, which uses AI to manage prior authorization decisions, is reportedly delaying care at Washington state hospitals. A U.S. senator called for the program to be scrapped after a new report documented the harm.
- Impact: The episode highlights a key risk in deploying AI for high-stakes administrative decisions in healthcare: when the model errs or slows decisions, real patients experience treatment delays. The controversy is likely to intensify congressional scrutiny of CMS AI initiatives.

2. Global AI Health Regulation: A Complex Patchwork
- What happened: A new analysis published this week examines how different regions — the EU, US, Australia, and China — are regulating AI in healthcare, largely under existing medical device frameworks. The piece notes significant divergence in implementation and enforcement.
- Impact: For multinational health AI companies, regulatory fragmentation creates compliance overhead and may slow market entry in key regions. The analysis underscores that no unified international standard yet exists.

3. Proposed Federal Rule of Evidence 707 Creates New AI Admissibility Questions
- What happened: A proposed new Federal Rule of Evidence 707, published April 23, would govern the admissibility of "machine-generated evidence" in federal court proceedings. Legal analysts say it creates novel challenges for practitioners relying on AI-generated clinical documentation, diagnostic outputs, or records.
- Impact: Healthcare organizations and health AI vendors face potential new legal exposure around AI-generated outputs used in litigation — including malpractice cases, insurance disputes, and regulatory proceedings.
Clinical Frontlines
American Hospital Association Forum — AI Leaders Discuss Governance and Policy Futures
- The AI: Senior hospital and health system leaders — including heads of Houston Methodist, Mass General Brigham, and the Joint Commission — convened at an AHA event on April 20 to discuss AI practices and policies across clinical and operational settings.
- Results: The forum addressed both the promise of AI in improving care delivery and the governance frameworks health systems need to deploy it responsibly. No single technology was highlighted; the focus was strategic adoption at scale.
- Significance: The participation of major health system CEOs signals that AI governance is now a board-level concern, not just an IT issue. The Joint Commission's involvement hints at potential future accreditation implications for AI deployment.

CancerNetwork — AI in Palliative Oncology for Hematologic Malignancies
- The AI: A new clinical review published April 22 explores how AI tools are being applied in palliative care for blood cancers, including applications in prognostication, symptom management, and personalizing end-of-life care pathways.
- Results: The review finds AI can meaningfully enhance prognostic accuracy in hematologic malignancies — a notoriously difficult domain — and help tailor symptom management for patients where curative treatment is no longer the goal.
- Significance: Palliative oncology has historically been underserved by AI research. This review positions AI as a tool for improving quality of life, not just survival outcomes — a meaningful expansion of the clinical use case.
Globenewswire — AI-Powered Intraoperative Nerve Monitoring Gains Momentum
- The AI: A market analysis published April 23 highlights AI-driven intraoperative neurophysiological monitoring (IONM) systems being deployed in spine, neurosurgical, and ENT procedures. These systems use machine learning to interpret nerve signals in real time, with remote monitoring capabilities filling gaps created by workforce shortages.
- Results: AI-enhanced IONM is reported to improve surgical precision and safety by providing real-time guidance that may not be available when specialist neurophysiologists are unavailable on-site.
- Significance: The combination of AI and remote monitoring addresses a genuine staffing crisis in surgical specialties, making it a commercially compelling and clinically relevant application poised for rapid adoption.
Funding & Deals
Earendil Labs — $787M (Largest Deal of Q1 2026)
- What they do: Deep learning drug discovery platform that has already generated 40+ therapeutic programs.
- Investors: Not disclosed in available reporting.
- Why it matters: The $787M raise is the single largest digital health deal of Q1 2026, signaling that AI-native drug discovery platforms are attracting mega-round funding on par with late-stage biotech. The volume of therapeutic programs already in the pipeline suggests investors are betting on platform breadth, not just a single drug candidate.
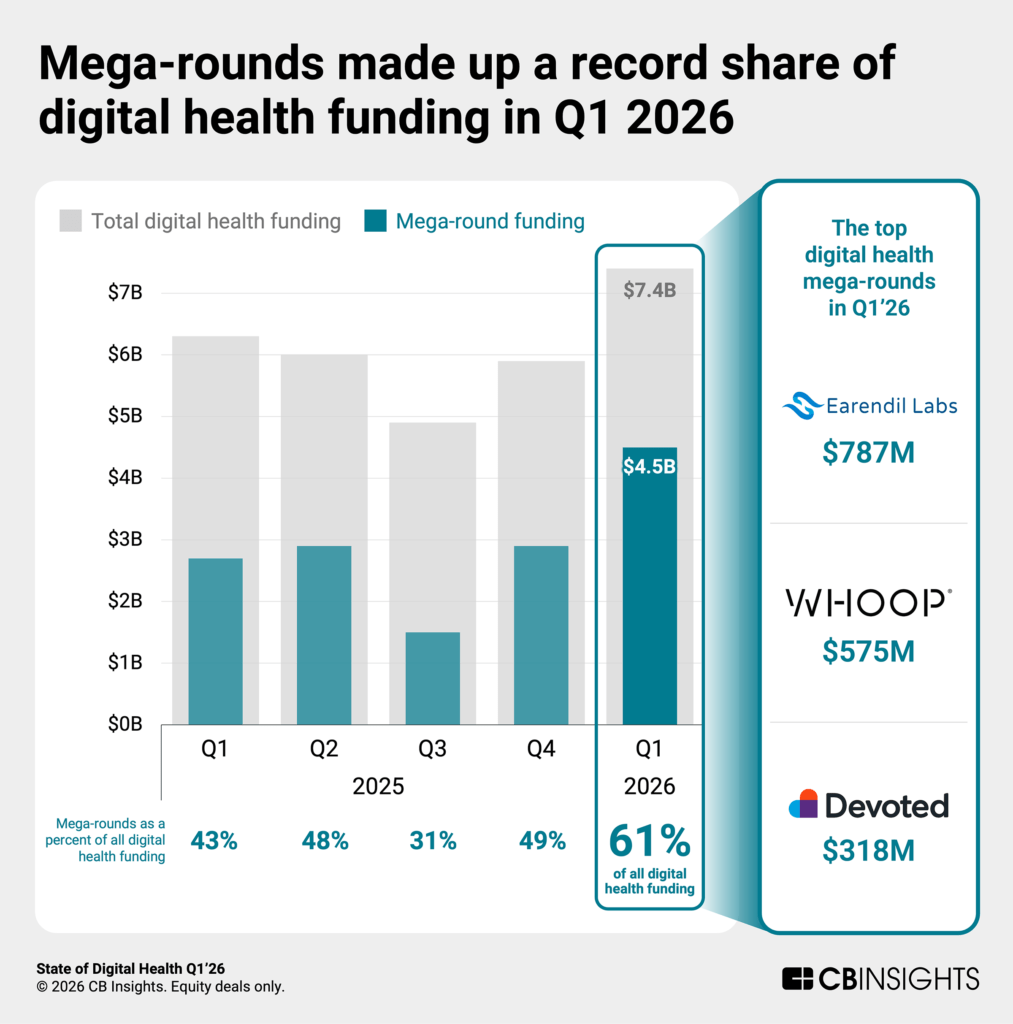
OpenEvidence — $250M Series D
- What they do: AI-backed medical information platform providing clinicians with evidence-based, real-time clinical decision support.
- Investors: Not disclosed in available reporting.
- Why it matters: A $250M Series D at this stage reflects strong commercial traction in clinical decision support — one of the most crowded but also highest-value segments of health AI. This raise positions OpenEvidence as a likely IPO or acquisition candidate.
Coral — $12.5M Seed
- What they do: Healthcare administrative automation startup focused on untangling complex workflows in billing, prior authorization, and care coordination.
- Investors: Lightspeed and Z47 led the round.
- Why it matters: With prior authorization delays making national headlines this week (see Regulatory section), Coral's raise is well-timed. Lightspeed's participation signals conviction in administrative AI as a durable category, not just a feature inside larger EHR platforms.
Qualified Health — $125M
- What they do: Enterprise platform that helps health systems evaluate and adopt AI technology responsibly.
- Investors: Not disclosed in available reporting.
- Why it matters: As hospitals face pressure to deploy AI quickly while managing governance risk, a neutral "AI evaluation layer" becomes strategically valuable. This raise confirms a market for AI infrastructure tools that sit between vendors and health systems.
Research Spotlight
"Is AI Actually Improving Healthcare?" — Nature Medicine
- Published in: Nature Medicine, Vol. 32 (April 2026); authored by Goldenberg, A. and Wiens, J.
- Key finding: The commentary directly challenges the field to produce rigorous evidence that AI deployments are translating into measurable patient benefit — not just algorithmic performance on benchmarks. The authors argue that claims of clinical improvement must be backed by appropriately designed evidence, including real-world outcomes data.
- Clinical relevance: This is a high-profile call for randomized or prospective evidence standards in clinical AI validation. Health systems and AI vendors deploying tools without outcome-level evidence may face growing scrutiny from payors, regulators, and clinicians emboldened by this framing.
"Utah's Prescription-Renewal Pilot Program — Autonomous AI Managing Patient Care" — NEJM
- Published in: New England Journal of Medicine, published April 18, 2026; authored by Ravi B. Parikh, M.D., M.P.P. (Emory University Department of Hematology and Medical Oncology).
- Key finding: The perspective examines Utah's pilot program in which AI autonomously manages prescription renewals for patients, raising fundamental questions about accountability, oversight, and the appropriate scope of AI agency in clinical care.
- Clinical relevance: This is among the first high-profile peer-reviewed discussions of fully autonomous AI in a direct patient-care workflow. It will likely inform debate about where the line should be drawn between AI as a decision-support tool and AI as a decision-maker — with implications for liability frameworks and regulatory policy.
What to Watch Next Week
- WISER program fallout: Expect CMS to respond to Senator pressure regarding the AI prior authorization pilot. Watch for congressional hearings or a formal program review announcement.
- Federal Rule of Evidence 707 comment period: Legal and health system stakeholders will begin weighing in on the proposed AI evidence admissibility rule. Healthcare attorneys should monitor docket activity closely.
- Nature Medicine editorial response: The "Is AI actually improving healthcare?" commentary is likely to generate replies and follow-on commentary from clinical AI researchers — watch for response letters and op-eds from major academic medical centers.
- Q1 2026 digital health funding follow-on analysis: Rock Health and other trackers are publishing full Q1 2026 analyses. Expect sector-specific breakdowns that will clarify where AI drug discovery and clinical AI are concentrating capital.
Reader Action Items
-
For health system leaders and compliance teams: The WISER program controversy and the proposed Federal Rule of Evidence 707 together signal a tightening legal and regulatory environment around AI-driven clinical decisions. Now is the time to audit which AI tools in your organization are making or influencing patient care decisions — and ensure human oversight protocols are documented and defensible.
-
For clinical AI vendors and investors: The Nature Medicine editorial demanding outcome-level evidence for AI's clinical value is a signal, not just a provocation. Products that can demonstrate randomized or prospective real-world evidence of patient benefit will be increasingly differentiated from those relying on benchmark performance alone. Build evidence generation into your roadmap.
-
For AI practitioners and researchers: The NEJM perspective on Utah's autonomous prescription-renewal AI raises questions the field must answer: What constitutes appropriate human oversight of autonomous AI in clinical workflows? Engage with this framing now — it will shape policy and procurement criteria within 12–18 months.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.