AI in Healthcare Pulse — 2026-05-05
This week in AI healthcare: the FDA launches a landmark real-time clinical trial pilot with AstraZeneca and Amgen, a Harvard study shows AI outperforming emergency room physicians at diagnosis, and NEJM AI spotlights a new generative chest X-ray model. Regulatory momentum is building fast, with clinical evidence now catching up.
AI in Healthcare Pulse — 2026-05-05
Regulatory & Policy Watch
1. FDA Launches AI-Powered Real-Time Clinical Trial Pilot with AstraZeneca and Amgen
- What happened: The FDA announced that cancer drug trials from AstraZeneca and Amgen will be monitored in real time using AI and cloud infrastructure — the first test of its kind aimed at shortening the interval between trial phases. A parallel Federal Register notice opened a public comment period on the broader AI-Enabled Optimization of Early-Phase Clinical Trials pilot program.
- Impact: The initiative could reduce 20–40% of overall clinical trial timelines, according to reporting. For pharma and biotech companies, this signals a major shift in how the FDA intends to engage with trial data — potentially accelerating drug approvals and reducing costs. Stakeholders have a formal window to provide input on expanding the program.

2. FDA Denies Harrison.ai Petition to Exempt Radiology AI from Premarket Review
- What happened: The FDA formally denied a citizen petition from Harrison.ai that had sought to exempt certain computer-aided detection (CAD) radiology AI devices from routine 510(k) premarket review. The petition had argued for greater flexibility in the clearance process for tools used by radiologists to analyze medical images.
- Impact: The denial reinforces the FDA's position that AI medical devices — even narrowly scoped diagnostic tools — require premarket oversight. This is a significant signal for AI radiology companies hoping to streamline their path to market.
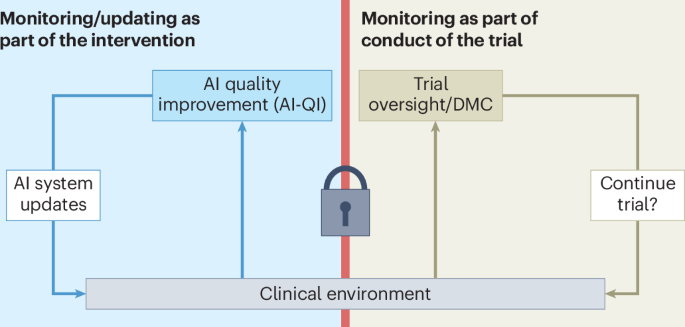
3. Nature Medicine Publishes Framework for Trialing Continuously Updated AI Systems
- What happened: A perspective published in Nature Medicine this week addressed the fundamental regulatory and scientific challenge of running clinical trials for AI systems that are continuously monitored and updated post-deployment — a category that existing trial frameworks were not designed to accommodate.
- Impact: As AI tools become embedded in clinical workflows and receive ongoing model updates, regulators and trial designers will need new standards for evidence generation. This paper provides a conceptual foundation that could inform FDA and international regulatory guidance going forward.
Clinical Frontlines
Harvard Medical School — AI Outperforms Emergency Room Physicians at Diagnosis
- The AI: Researchers tested OpenAI's o1-preview large language model — among others — on real emergency room cases, evaluating diagnostic accuracy and clinical decision-making.
- Results: At least one AI model demonstrated higher diagnostic accuracy than two human emergency physicians working on the same cases. Researchers described the results as marking "a really profound change in technology that will reshape medicine." Even the study authors were reported to be surprised: "We're already at the ceiling," according to Fortune.
- Significance: This is among the most rigorous real-world evaluations of LLM diagnostic performance to date, conducted on actual ER cases rather than medical licensing exam questions. The findings intensify the debate about how AI should be integrated into emergency triage and whether clinical AI can now serve as a co-equal decision-support tool rather than just a background assistant.

WBUR / Massachusetts Hospitals — AI Diagnostic Tools Enter Doctor's Offices Across the State
- The AI: A range of AI-powered diagnostic tools, from CT scan enhancement software to generative AI chatbots that analyze patient records, are now in active use across Massachusetts hospitals and physician practices.
- Results: Reporting from WBUR (published May 4) describes AI tools becoming a routine fixture of clinical encounters in the state, with physicians using AI to analyze imaging, review patient histories, and support triage decisions.
- Significance: This represents a meaningful inflection point: AI-assisted care is no longer a pilot or an experiment in leading academic medical centers — it is entering everyday clinical practice at scale across a major state.

AAAS Science — Independent Analysis Confirms AI's Edge in Clinical Decision-Making
- The AI: Large language models evaluated in a simulated emergency room environment.
- Results: Science (AAAS) reported this week that AI systems are "starting to beat doctors at making correct diagnoses," corroborating the Harvard study findings with independent analysis. The piece emphasized performance in fast-paced, simulated ER conditions.
- Significance: Multiple independent outlets covering the same underlying research signals that AI diagnostic superiority in at least some clinical contexts is becoming harder to dismiss as an outlier finding. Clinicians and health systems will face increasing pressure to define their policies on AI-assisted diagnosis.

Funding & Deals
Yuzu Health — $35M (Series Not Disclosed)
- What they do: Behavioral and mental health platform incorporating AI-driven care navigation and clinical support tools.
- Investors: Details reported in Fierce Healthcare's 2026 fundraising tracker; specific lead investors not disclosed in available data.
- Why it matters: Mental health remains one of the most active verticals in digital health AI, and this raise signals continued investor appetite even as broader funding markets remain selective.

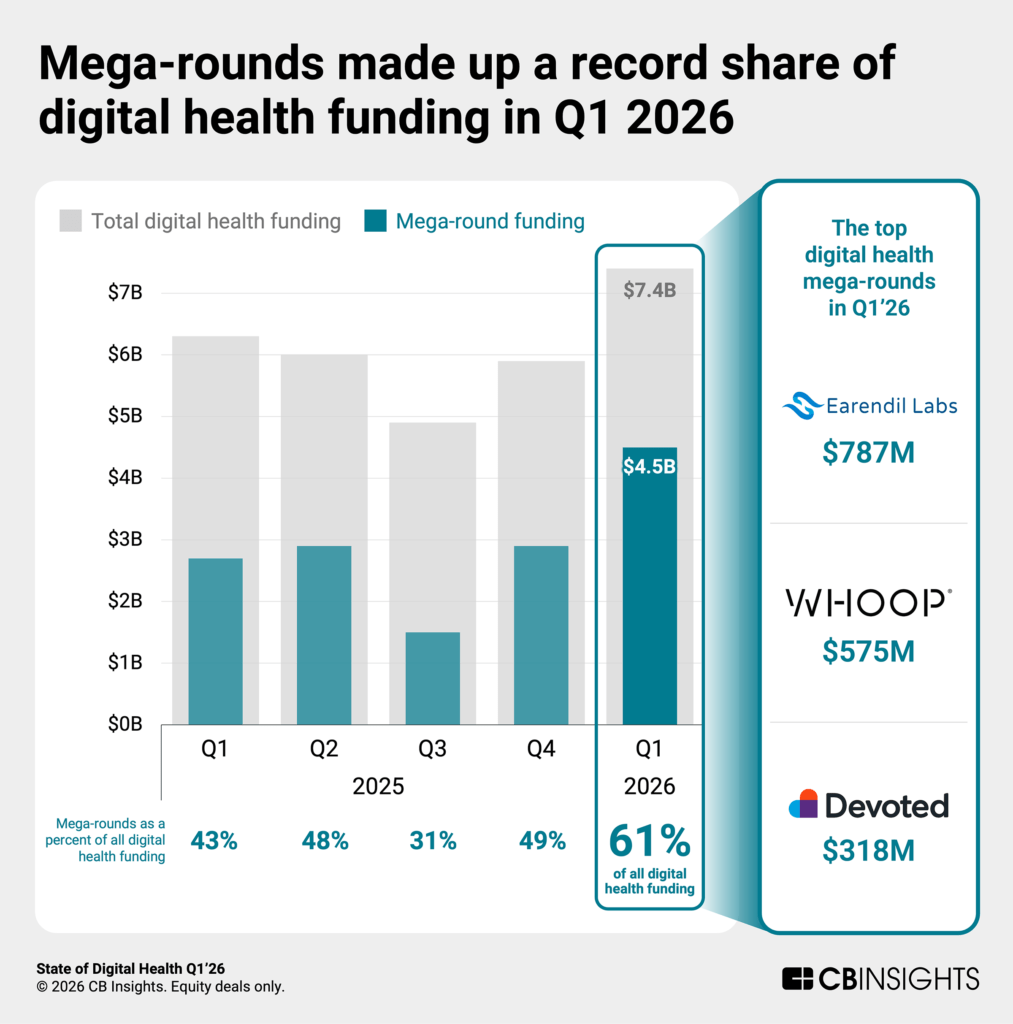
Digital Health Sector — $7.4B Raised in Q1 2026, AI Drug Discovery Leads
- What they do: Aggregate sector-level data from HIT Consultant tracking AI-driven drug discovery platforms and digital health M&A.
- Investors / Deals: Earendil Labs raised $787M — the largest single deal of the quarter — for a deep learning platform that has already generated 40+ therapeutic programs. Takeda committed up to $1.7B to Iambic Therapeutics, an AI drug discovery firm.
- Why it matters: Q1 2026 digital health funding totaled $7.4B, driven by AI-enabled drug discovery megadeals. This dwarfs the $4B Rock Health figure for the same period, reflecting how differently large pharma–AI partnerships are counted. Either way, the trend is clear: institutional capital is flowing heavily into AI-first drug development platforms.
Health Universe — $6M Seed
- What they do: A platform for integrating AI into healthcare organizations — serving as a marketplace and deployment layer for clinical AI tools.
- Investors: Led by Kleiner Perkins, with participation from Susa Ventures, Twelve Below, and Oncology Ventures.
- Why it matters: Kleiner Perkins leading a health AI infrastructure seed round underscores growing investor interest not just in point-solution AI tools, but in the deployment and integration layer that lets hospitals and health systems actually operationalize AI at scale.
Research Spotlight
"Is AI Actually Improving Healthcare?" — Nature Medicine Perspective
- Published in: Nature Medicine (Vol. 32, 2026)
- Key finding: Authors Goldenberg and Wiens challenge the field to move beyond benchmark performance and ask whether AI tools are demonstrably improving health outcomes in real-world deployments. The piece calls for rigorous outcome-focused evidence rather than model accuracy metrics alone.
- Clinical relevance: As AI tools proliferate in clinical settings, this perspective serves as a critical counterweight to hype — pushing health systems, regulators, and developers toward outcome-based evaluation frameworks. Particularly timely given the wave of ER diagnostic AI results published this week.
NEJM AI — ChexGen: Generative Vision-Language Foundation Model for Chest X-Rays
- Published in: NEJM AI
- Key finding: ChexGen is introduced as a generative vision-language foundation model providing a unified framework for synthesizing chest X-ray images guided by text descriptions, masks, or bounding boxes. The model enables controlled, clinically meaningful image generation across multiple chest pathology categories.
- Clinical relevance: Generative chest imaging models like ChexGen have several potential clinical applications: augmenting training datasets for rare conditions, supporting radiologist education, and enabling privacy-preserving synthetic data for AI model development. If validated in downstream diagnostic tasks, such models could meaningfully improve the robustness of radiology AI across underrepresented patient populations.
What to Watch Next Week
- FDA RFI comment window: The Federal Register notice on the AI-Enabled Early-Phase Clinical Trials pilot program is open for public input. Expect responses from pharma, biotech, and patient advocacy groups that will shape how the pilot is designed and expanded.
- Harvard ER AI study peer-review publication: The underlying research driving this week's headlines is expected to be formally peer-reviewed and published. The full methods and dataset details will clarify how generalizable the findings are across different hospitals and patient populations.
- Regulatory posture on AI diagnostics: With AI systems now demonstrably matching or exceeding physician performance in emergency settings, watch for statements from the FDA, AMA, and hospital systems on policy responses — particularly around liability, disclosure to patients, and required human oversight.
- Q2 digital health funding data: With Q1 figures confirmed at $4–7.4B (depending on methodology), early Q2 deal flow will indicate whether the AI-drug-discovery mega-deal pace is sustainable or a one-quarter spike.
Reader Action Items
-
Clinicians and hospital administrators: The Harvard ER AI diagnostic study is a must-read for anyone developing or reviewing AI policy in your institution. The results shift the baseline assumption — AI is no longer clearly inferior to physicians in triage and diagnosis. Begin scoping an internal policy on when and how AI diagnostic tools should be used, disclosed to patients, and integrated with physician oversight.
-
Healthcare AI developers: The FDA's denial of the Harrison.ai petition, combined with its simultaneous launch of a real-time trial pilot, signals a nuanced regulatory posture: the FDA is open to accelerating AI-enabled drug development but is not relaxing premarket review requirements for medical device AI. Engage with the Federal Register comment process to shape the pilot program before it becomes a template.
-
Investors: Earendil Labs ($787M) and Iambic Therapeutics (up to $1.7B from Takeda) define the upper end of this market. But infrastructure plays — like Health Universe's Kleiner-led seed — may offer better near-term return profiles as the deployment bottleneck, not model quality, becomes the rate-limiting factor for health AI adoption.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.