AI in Healthcare Pulse — 2026-04-28
This week in AI healthcare: a new national AI legislative framework signals a federal shift on healthcare AI regulation, AWS publishes a guide on multimodal biological foundation models for drug discovery, and Nature Medicine releases back-to-back commentary on whether clinical AI is actually delivering results. Fresh funding data shows Q1 2026 was the strongest digital health quarter since the pandemic peak.
AI in Healthcare Pulse — 2026-04-28
Regulatory & Policy Watch
1. 2026 National AI Legislative Framework — Healthcare Implications
- What happened: A new national AI legislative framework has emerged with significant implications for healthcare. According to legal analysis published this week, the framework signals a move toward federal AI legislation that could preempt state-level laws, creating a more unified regulatory environment for health AI developers and deployers.
- Impact: Healthcare AI companies operating across multiple states currently navigate a patchwork of state regulations. A federal preemption regime would simplify compliance but may also raise the floor on requirements across the board — particularly for liability and transparency. Health systems and AI vendors should monitor congressional developments closely.

2. LA Times Op-Ed: AI Could Democratize Medicine — But Regulation Must Come First
- What happened: UCLA emergency medicine physician Dr. Hashem Zikry published a commentary in the Los Angeles Times arguing that AI has the potential to significantly expand healthcare access for patients facing doctor shortages, high costs, and long wait times — but only if proper regulatory guardrails are established first.
- Impact: The commentary reflects a growing clinical consensus: AI's promise for democratizing healthcare is real, but deployment without oversight risks patient harm. The piece adds a clinician's voice to ongoing federal and state-level debates about how aggressively to regulate AI-assisted care.
3. Nature Medicine Calls for Clearer Evidence Standards from Regulators
- What happened: A commentary published in Nature Medicine this week titled "Show us the evidence for the value of medical AI" argues that regulators should better clarify which categories of medical AI tools require prospective evidence of clinical impact versus those that can enter practice under more limited claims.
- Impact: The piece directly challenges healthcare organizations and administrators to distinguish among pilot implementation, operational use, and evidence of benefit — rather than treating these as a single decision. For the FDA and CMS, this represents a call to build tiered evidentiary frameworks into their AI device oversight approach.
Clinical Frontlines
AWS — Multimodal Biological Foundation Models Across Drug Discovery and Patient Care
- The AI: AWS published a deep-dive this week on multimodal biological foundation models (BioFMs) — large AI systems trained on multiple biological data types (genomic, proteomic, imaging, clinical) simultaneously. The post details real-world applications across drug discovery, clinical development, and patient care workflows.
- Results: AWS highlights that organizations using multimodal BioFMs can accelerate therapeutic program generation, with the company showcasing its infrastructure for deploying these models at scale.
- Significance: This marks a maturation point for AI in drug discovery — moving from single-modality models (e.g., protein folding only) to integrated systems capable of spanning the full therapeutic pipeline. Cloud-scale deployment lowers the barrier for mid-size biotech and academic medical centers to access these capabilities.
Nature Medicine — Is Clinical AI Actually Improving Healthcare?
- The AI: A commentary by Goldenberg and Wiens published in Nature Medicine (Vol. 32) this week asks a blunt question: Is AI actually improving healthcare? The piece surveys the current state of evidence across clinical AI deployments.
- Results: The authors do not offer a blanket verdict, but the framing of the question reflects growing concern in the research community that AI tools are being widely deployed without rigorous evidence of benefit.
- Significance: Coming from Nature Medicine, this signals that even AI-friendly academic communities are demanding higher evidentiary standards before clinical AI can be considered proven. Developers and investors should expect increasing pressure for randomized trial data.
Big Pharma Meets Big Tech — AI Bolsters Drug Discovery Pipeline
- The AI: A report published this week by IBTimes examines how pharmaceutical companies are embedding AI into drug discovery pipelines, increasingly blurring the boundary between traditional pharma and technology companies.
- Results: The piece describes how AI is compressing drug development timelines and enabling novel target identification across the industry.
- Significance: As AI becomes a core component of pharmaceutical R&D, regulatory bodies will need to address how AI-generated drug candidates are validated and approved — an area where current frameworks still have significant gaps.
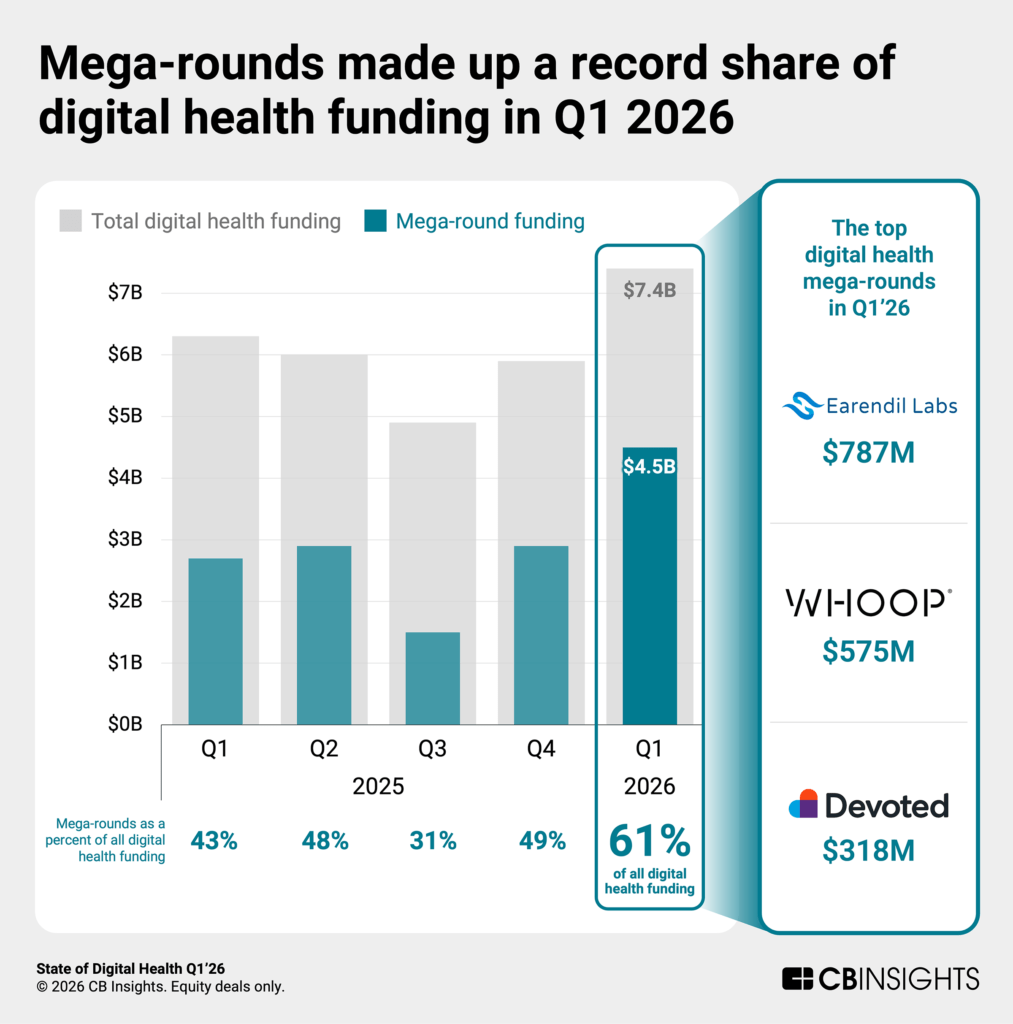
Funding & Deals
Earendil Labs — $787M (Q1 2026 Mega-Round)
- What they do: Earendil Labs operates a deep learning platform for therapeutic program generation, with 40+ therapeutic programs already generated by its AI system.
- Investors: Not disclosed in the available reporting.
- Why it matters: This was the single largest deal of Q1 2026 in digital health, according to HIT Consultant's analysis of the quarter. The size of the round signals that AI-native drug discovery — not just AI-assisted discovery — is now commanding venture-scale capital commitments.
Takeda + Iambic Therapeutics — Up to $1.7B Partnership
- What they do: Iambic Therapeutics uses AI-driven drug design to accelerate small molecule therapeutics from discovery through clinical development.
- Investors / Partner: Takeda Pharmaceuticals committed up to $1.7 billion to the partnership.
- Why it matters: This is one of the largest AI-drug discovery deals on record, and reflects a broader trend of major pharma betting heavily on AI platforms rather than building capabilities in-house. The Takeda-Iambic partnership also demonstrates that "timeline compression" — getting drugs to clinical stages faster — is the primary commercial value proposition AI is delivering to pharma.
Digital Health Sector — $4B Raised in Q1 2026
- What they do: Across 110 deals, digital health startups collectively raised $4 billion in Q1 2026 — $1 billion more than the same period in 2025 and the strongest first quarter since the pandemic peak, per Rock Health data.
- Investors: Broad venture participation across the sector.
- Why it matters: The rebound confirms that investor confidence in health tech has recovered and is now being channeled toward AI-native companies in drug discovery, diagnostics, and clinical workflow automation. AI is described as "table stakes" in current digital health pitches, according to Fierce Healthcare's coverage.
Research Spotlight
"Is AI Actually Improving Healthcare?" — Nature Medicine
- Published in: Nature Medicine, Vol. 32 (April 2026)
- Key finding: Goldenberg and Wiens argue that despite widespread adoption, the evidence base for clinical AI improving patient outcomes remains thin. The commentary calls for systematic evaluation of AI tools in real-world care settings.
- Clinical relevance: For hospital administrators and clinical informaticists currently deploying or evaluating AI tools, this piece serves as a caution: operational adoption should not be conflated with clinical validation. The authors push for prospective, outcome-focused evidence before AI tools are embedded in standard care pathways.
"Show Us the Evidence for the Value of Medical AI" — Nature Medicine
- Published in: Nature Medicine, Vol. 32 (April 2026)
- Key finding: This commentary argues that regulators, healthcare organizations, and AI developers each have distinct responsibilities in the evidence generation chain. Regulators should clarify evidentiary tiers; organizations should not collapse pilot and production decisions; developers should support rather than resist clinical trial requirements.
- Clinical relevance: The piece has direct implications for the FDA's ongoing discussions about tiered oversight of AI devices — and for the growing number of health systems that have moved AI tools from pilot to routine clinical use without clear outcome data.
What to Watch Next Week
- Federal AI preemption debate: The newly analyzed 2026 national AI legislative framework is still early-stage — watch for Congressional hearings or CMS/FDA guidance responses that could clarify how federal standards interact with state healthcare AI laws.
- Big Pharma AI partnerships: Following the Takeda-Iambic deal, watch for additional large-cap pharma companies announcing AI platform partnerships — Q2 2026 is shaping up to be active for this deal type.
- Evidence standards pressure: With two Nature Medicine commentaries this week calling for stronger clinical evidence, expect medical societies and hospital networks to begin issuing internal AI governance policies that require prospective validation before deployment.
- Multimodal BioFM adoption: AWS's public tutorial on deploying biological foundation models signals that cloud infrastructure for AI drug discovery is now commoditized. Watch for smaller biotech firms and academic medical centers announcing BioFM partnerships in coming weeks.
Reader Action Items
-
For health system AI leaders: Before expanding any AI tool from pilot to full clinical deployment, establish clear metrics distinguishing operational performance from clinical outcome improvement — the Nature Medicine commentaries this week make clear that conflating these creates both scientific and regulatory risk.
-
For healthcare investors: Q1 2026's $4B digital health raise is a signal, not a guarantee. Prioritize deals where companies can demonstrate prospective clinical evidence — the gap between AI that performs well in benchmarks and AI that improves patient outcomes remains the sector's defining risk.
-
For compliance and regulatory teams: Begin mapping your AI product portfolio against the emerging federal AI legislative framework analyzed this week. If federal preemption of state AI laws advances, companies with multi-state deployments will need to reassess compliance workflows — starting that analysis now puts you ahead of the curve.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.