Genetics & Genomics Frontiers — 2026-04-28
This week's most significant genetics news centers on a landmark Phase 3 trial success for Intellia Therapeutics' CRISPR-based hereditary angioedema treatment, while new research reveals a vast Indigenous American genome map exposing previously unknown migrations and over a million new genetic variants. The FDA's "Plausible Mechanism Framework" continues to reshape how ultra-rare gene editing therapies reach patients, with fresh analysis published this week.
Genetics & Genomics Frontiers — 2026-04-28
Key Highlights
Intellia's CRISPR Treatment Clears Phase 3 Trial
Intellia Therapeutics announced that its CRISPR-based treatment for hereditary angioedema met its primary endpoints in a Phase 3 clinical trial — a historic milestone for the gene editing field. This marks one of the first CRISPR therapies to succeed at the Phase 3 stage, moving the technology closer to broad clinical use.

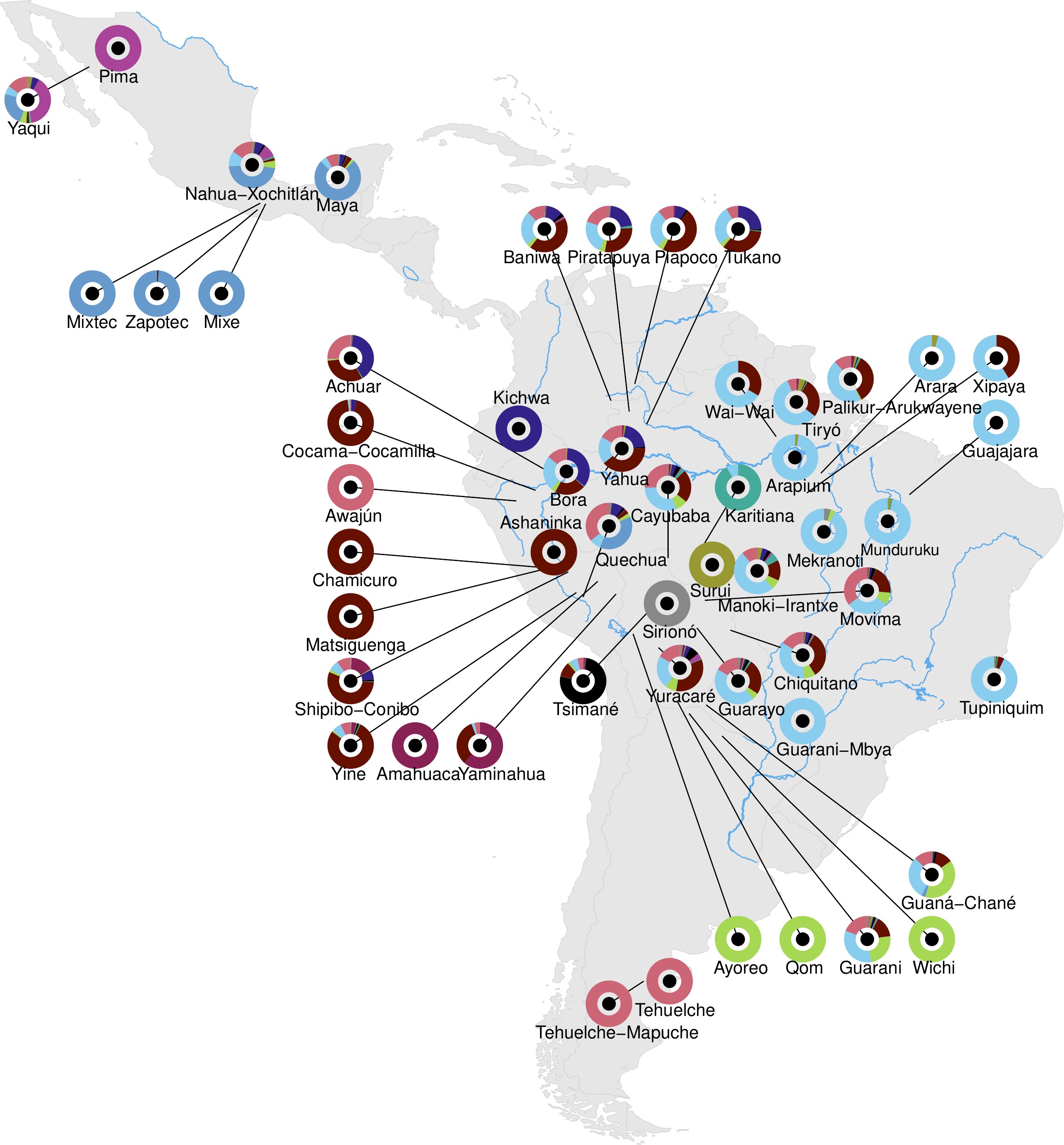
Indigenous American Genome Map Uncovers Lost Migrations
A sweeping new genomic analysis has produced a vast Indigenous American genome map, revealing previously unknown ancient migrations, over one million new genetic variants, and evidence of complex ancestral relationships. The study identified three major genomic groupings and detected approximately 2% Australasian-related ancestry and 1–3% archaic hominid ancestry among certain populations. Researchers also found genomic signatures of natural selection affecting immune function and metabolic traits.
FDA's Plausible Mechanism Framework: New Analysis
Fresh analysis published this week examines how the FDA's "Plausible Mechanism Framework" is accelerating individualized gene therapy review for ultra-rare diseases. The framework prioritizes biological proof of mechanism over large-scale clinical trials — a shift that could open the door to bespoke treatments for patients with conditions too rare to support conventional randomized trials.

Johns Hopkins: CRISPR as DNA's "Copyeditor"
A feature published this week by Johns Hopkins University's magazine reflects on the landmark first use of CRISPR to cure a life-threatening disease, framing the technology as medicine's new molecular "copyeditor." The piece contextualizes the rapidly accelerating transition from reading DNA to rewriting it in real clinical settings.

Analysis
The most promising development this week is unquestionably Intellia Therapeutics' Phase 3 success with its CRISPR-based hereditary angioedema treatment. While CRISPR has achieved earlier clinical milestones — including the approved sickle cell disease therapy and a widely publicized patient-specific base editing case — Phase 3 success represents the rigorous, large-scale validation that regulators and the broader medical community require before a therapy can become standard of care.
This milestone arrives as the regulatory environment is also evolving in favor of gene editing innovation. The FDA's Plausible Mechanism Framework is already enabling faster review for ultra-rare disease therapies, and late-stage CRISPR programs in transthyretin amyloidosis and hereditary angioedema are now both in advanced stages of development. Together, these converging forces — clinical success, supportive regulation, and maturing trial infrastructure — suggest 2026 may mark a genuine inflection point for CRISPR as a clinical modality.
What to Watch
- Intellia Therapeutics regulatory pathway: Following the Phase 3 success in hereditary angioedema, watch for regulatory filing timelines and potential FDA fast-track or priority review designations.
- CRISPR Therapeutics Phase 2 cholesterol trial: Phase 2 studies of CRISPR Therapeutics' cholesterol-lowering gene editing therapy were planned to begin in 2026, targeting broader patient populations and longer-term outcomes.
- Indigenous American genomics follow-up studies: The newly released genome map is expected to catalyze further research into population-specific disease variants and pharmacogenomics relevant to Indigenous communities.
- FDA NGS genome editing safety guidance: The FDA's draft guidance on safety assessment of genome editing in gene therapy products using next-generation sequencing remains open for industry comment — watch for final guidance publication.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.