Medical Device and Healthcare Industry Trends — 2026-04-21
This health signal was created by a user. It may contain unverified medical claims. Always consult a qualified healthcare professional before making health decisions.
South Korea's digital healthcare AI industry is entering a serious monetization phase, with Lunit hitting $60 million in annual revenue and other players gaining global traction. As the Ministry of Food and Drug Safety (MFDS) refines its innovative medical device fast-track and strengthens GMP audits, domestic firms are adjusting their regulatory strategies. Meanwhile, the FDA’s firm stance on AI medical device regulation has become a critical factor for Korean MedTech companies eyeing the U.S. market.
Medical Device and Healthcare Industry Trends — 2026-04-21
Top 5 Headlines of the Day
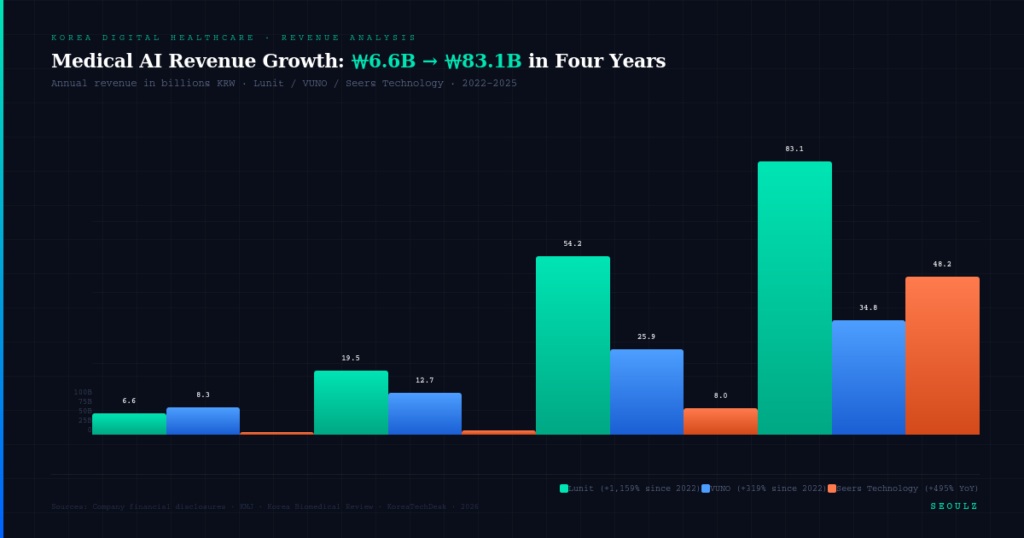
- Korean Medical AI Commercialization Begins: Lunit surpasses $60M in annual revenue, Seers turns profitable — K-Medical AI attracts global investors.
- Legal Clash at Samsung Semiconductor: Samsung Electronics files for an injunction to prevent labor union occupation of facilities — Potential impact on the operational stability of medical device subsidiaries.
- Philips AI CT System Wins FDA Approval: Aiming to increase premium CT market share by 2028 — Direct competition with Korean imaging firms like Samsung Medison.
- FDA Rejects AI Regulation Loosening: Decision made to maintain oversight on high-risk AI devices — Korean firms must re-evaluate U.S. approval strategies.
- Cala Health Gets FDA Approval for Wearable Neuromodulation Device: kIQ Plus cleared for essential tremor and Parkinson's disease — Global competition in therapeutic wearables accelerates.
Key Corporate Developments (4 Cases)
Lunit — Leading the Korean Medical AI Revenue Era
- What happened: Lunit reported over $60 million in annual revenue, marking the first time a Korean digital healthcare firm has entered a full-scale monetization stage. They also partnered with CellCarta for companion diagnostics in digital pathology AI.
- Numbers: $60M in annual revenue.
- Implications: Viewed as the first success story of a Korean medical AI company achieving real revenue through clinical evidence and global partnerships. Competitors like Vuno and Seers are also pushing for profitability, raising the overall investment appeal of the Korean medical AI sector.
Samsung Medison — Launch of 'Samsung HME America'
- What happened: Samsung Medison has integrated its two U.S. medical imaging entities, Neurologica and Boston Imaging, into 'Samsung HME (Healthcare and Medical Equipment) America'. This is a strategic move to centralize U.S. investment and R&D capabilities.
- Numbers: 2 entities merged into 1 (Announced March 23, 2026).
- Implications: Seen as a push to compete more effectively with global giants like Philips, Siemens, and GE by ensuring brand consistency and operational efficiency in the U.S. market.
Genbody — Chasing Korea's First HIV Home Diagnostic Kit Approval
- What happened: In line with the MFDS policy to expand at-home diagnostic kits, Genbody is aiming for domestic approval of the first HIV home test by mid-2025.
- Numbers: Targeting the new HIV self-testing market (would be the first in Korea).
- Implications: The expansion of self-testing items by the MFDS creates new domestic revenue streams. Other diagnostic leaders like SD Biosensor and Humasis are expected to expand their at-home product lineups.

Lunit × CellCarta — AI Digital Pathology Companion Diagnostics Partnership
- What happened: Lunit partnered with global companion diagnostics firm CellCarta to accelerate the adoption of AI-based digital pathology workflows, specifically for cancer drug development.
- Numbers: Partnership signed — deal size undisclosed.
- Implications: Companion diagnostics is a high-growth field. This partnership embeds Lunit’s AI pathology solutions directly into global clinical pipelines, signaling the integration of Korean AI firms into the global clinical ecosystem.
MFDS, Policy, and Regulation (3 Cases)
Innovative Medical Device 'Immediate Market Entry' Fast-Track
- Details: The Ministry of Health and Welfare and the MFDS officially launched the 'Market Immediate Entry Medical Technology' system on January 26, 2026, creating a rapid market access pathway for AI and big data-driven medical devices.
- Target: Medical devices applying innovative technologies like AI and big data.
- Effect: Following a 50% jump in innovative medical device designations in 2025, this system is expected to shorten time-to-market for domestic firms. The next challenge is bridging the gap between MFDS approval, insurance coverage, and hospital adoption.
Abolition of 'Sales Suspension Grace Period' for GMP Violations
- Details: The MFDS has strengthened compliance for in-vitro diagnostic (IVD) GMP renewals, banning imports and sales during the 'grace period' that was previously permitted.
- Target: All IVD manufacturers and importers.
- Effect: Small-to-medium firms that neglect GMP updates face immediate sales suspension risks. Systematic schedule management is now a core competitive factor.
Expanding Scope of Home Diagnostic Kits — Includes Flu, STD, and Drugs
- Details: The MFDS announced a plan to significantly expand the scope of self-testing kits to include influenza, STDs, and narcotics.
- Target: IVD manufacturers, especially those making self-test kits.
- Effect: This marks a structural shift from hospital-centric diagnostics to consumer-facing home testing, with firms like Genbody, SD Biosensor, and Seegene expected to ramp up B2C entry.
Digital Health & AI Medical (3 Cases)
- Lunit: Surpassed $60M in annual revenue and partnered with CellCarta for global clinical expansion.
- Seers: Successfully turned a profit, proving the viability of its AI medical solution business model.
- Philips (AI CT System): FDA approval secured; aiming to expand in the premium CT market by 2028, intensifying global competition with Samsung Medison and others.

Global MedTech Context
- FDA Rejects AI Loosening (April 9, 2026): The decision to maintain strict oversight on high-risk AI means Korean firms like Lunit and Vuno must prioritize rigorous clinical data and regulatory strategy.
- Philips AI CT FDA Approval: Intensifies competition with Samsung Medison; underscores the urgent need for product differentiation and advanced AI features.
- Cala Health kIQ Plus Approval: With new wearable neuro-stimulation FDA clearances, domestic startups targeting neurological diseases must carefully assess their technical gap with global leaders.
Investment, M&A, and Listing Trends
- GI Vita Series A: Secured 4.5 billion KRW in Series A funding to expand digital health services for pre-diabetic patients.
- Sector-wide Monetization: Global investor interest in Korean medical AI is peaking due to the financial milestones of Lunit and Seers, likely leading to more investment rounds and overseas listing attempts.
Today's Insight
The Korean medical AI industry is at a historic turning point, shifting from "technology demonstration" to "revenue generation." The financial milestones achieved by Lunit and Seers validate the business model of clinical-evidence-based AI. While the MFDS provides a solid domestic foundation through fast-track and self-testing policies, the FDA’s strict regulatory stance and the aggressive moves by global incumbents like Philips remind Korean firms that clinical rigor remains non-negotiable.
Checkpoints for This Week
- Confirm final schedule for MFDS self-test kit regulation updates.
- Monitor Q1 earnings and potential global partnership announcements from Lunit, Vuno, and others.
- Observe the Samsung Electronics labor union injunction case and its impact on medical device subsidiaries.
Action Items for Readers
- Regulatory/Compliance: Review the latest MFDS official notices regarding 'Market Immediate Entry' fast-track and strengthened GMP audits to see if they apply to your product pipeline.
- Investors: Use Lunit and Seers as benchmarks to evaluate the revenue-generation timelines and partnership status of Korean medical AI firms in your portfolio.
- Startup Founders: Take the FDA's decision to maintain strict AI oversight into account; adjust your U.S. entry strategy conservatively and prioritize collecting high-quality clinical evidence early.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.