Pandemic & Infectious Disease — 2026-04-28
WHO wrapped up Exercise Polaris II, a landmark two-day simulation involving 26 countries and 600 health emergency experts, aimed at stress-testing global response capacity to a fictional novel bacterium outbreak. On the vaccine front, the FDA approved Bavarian Nordic's chikungunya vaccine for ages 12 and older, while Butantan's single-dose dengue vaccine demonstrated 80.5% protection against severe dengue over five years in a Phase 3 trial. Meanwhile, STAT News experts are reassessing COVID-19's trajectory, noting severity has waned significantly even as questions remain about who still benefits from booster shots.
Pandemic & Infectious Disease — 2026-04-28
Active Outbreak Tracker
Exercise Polaris II: Novel Pathogen Simulation — Global
- Status: Concluded April 27, 2026; exercise wrapped successfully after two days
- Key Development: WHO convened 26 countries and territories, over 600 health emergency experts, and more than 25 international partners for Exercise Polaris II — a high-level simulation built around an outbreak of a fictional novel bacterium spreading worldwide. The exercise is designed to identify gaps in coordinated pandemic response before a real emergency strikes.
- Response: WHO and partner governments engaged in tabletop and operational drills across response pillars including surveillance, communications, logistics, and international coordination. Findings are expected to inform future preparedness planning.

Respiratory Viruses (COVID-19, Flu, RSV) — United States
- Status: Activity declining; RSV declining or likely declining in 48 U.S. states as of April 21, 2026; no states showing RSV growth
- Key Development: CDC's Center for Forecasting and Outbreak Analytics (CFA) confirmed that as of April 21, 2026, RSV infections are growing or likely growing in 0 states, declining or likely declining in 48 states, and not changing in 1 state. The CDC noted it does not anticipate producing additional seasonal outlook updates for the remainder of the 2025–2026 respiratory season, signaling the season is winding down.
- Response: Routine surveillance continues. CDC is monitoring respiratory disease activity across all U.S. states and updating Rt estimates for COVID-19, influenza, and RSV.
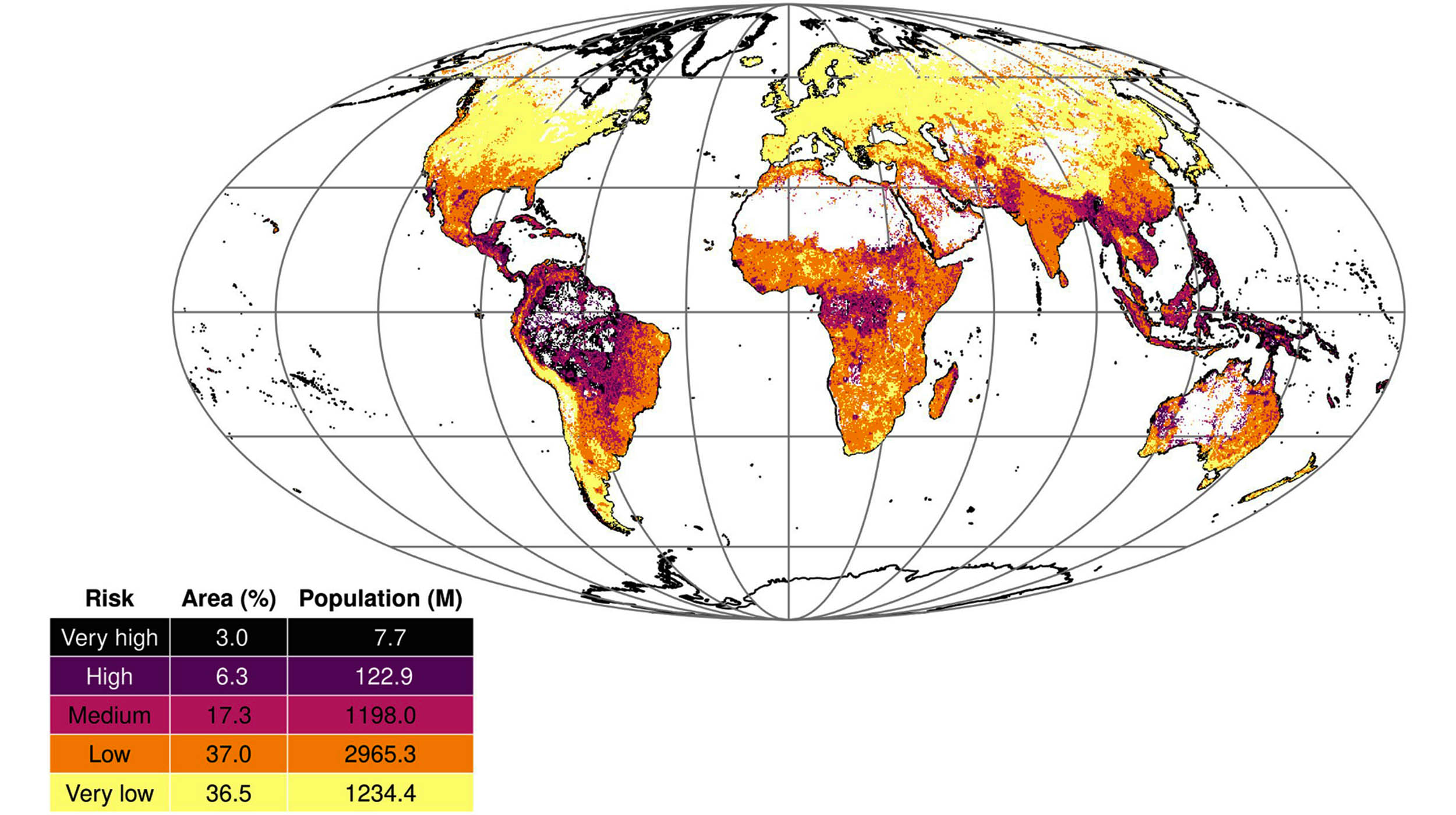
Disease Outbreak Vulnerability — Global Hotspots
- Status: New global modeling identifies 9.3% of the world's land area as "highly vulnerable" to dangerous disease outbreaks
- Key Development: A newly published global disease outbreak risk map, released within the past week, flags regions most at risk of harboring and spreading dangerous pathogens. The modeling synthesizes ecological, demographic, and health system factors to produce a global vulnerability index.
- Response: The mapping tool is intended to guide international preparedness investments and outbreak surveillance prioritization.
Vaccine & Treatment Pipeline
-
Chikungunya Vaccine (Bavarian Nordic): FDA approved Bavarian Nordic's chikungunya vaccine for individuals aged 12 and older, based on positive Phase 3 clinical trial results. The approval offers travelers and others at risk of the mosquito-borne illness an effective option.
-
Butantan-DV Dengue Vaccine (Butantan Institute): A Phase 3 clinical trial found that the Butantan-DV tetravalent single-dose dengue vaccine provided 80.5% protection against severe dengue with warning signs over five years and prevented hospitalizations among vaccinated participants — a significant durable efficacy milestone for a once-a-lifetime dosing strategy.

-
COVID-19 Vaccine 2026–2027 Formula (FDA/VRBPAC): The FDA published a Federal Register notice announcing a forthcoming Vaccines and Related Biological Products Advisory Committee (VRBPAC) meeting to deliberate on the U.S. COVID-19 vaccine antigen composition for the 2026–2027 season. A public docket has been established for comments.
-
Lyme Disease Vaccine Candidate PF-07307405/LB6V (Pfizer): A Phase 3 trial demonstrated "strong efficacy" exceeding 70% for Pfizer's Lyme disease vaccine candidate. Pfizer has announced the candidate and is expected to pursue regulatory filings based on this trial success.
-
Single-dose malaria medicine, bird flu vaccine, HIV treatment (Multiple): The Global Health Technologies Coalition's April 27 research roundup highlighted three notable developments: a single-dose malaria medicine rollout, progress on a bird flu vaccine trial, and an HIV treatment approval — underscoring a packed week of infectious disease pipeline news.

Expert Analysis
COVID-19 has evolved significantly since its pandemic peak, and experts are now reassessing its trajectory with nuance. A STAT News analysis published April 27 gathered perspectives from multiple epidemiologists examining why COVID severity has declined, who still benefits from booster shots, and whether the virus now behaves more like a common cold or seasonal flu. The consensus emerging is cautious: population immunity, improved treatments, and variant evolution have collectively blunted the disease's worst outcomes — but COVID has not become trivial, particularly for the elderly, immunocompromised individuals, and those with underlying conditions.

The debate over vaccine recommendation pathways in the U.S. remains unresolved. A Reuters report confirmed that Americans may face the upcoming respiratory disease season without clear guidance on COVID-19 shots and updated flu vaccines, following a federal court ruling that halted the Kennedy-era advisory panel. The ruling has left several vaccines in limbo and raised questions about whether newly approved products will be covered by insurers — a policy vacuum with direct public health implications.
On the pandemic preparedness front, WHO's Exercise Polaris II is being viewed by public health analysts as a critical test of whether lessons from COVID-19 have been operationalized. Bringing together 600 experts from 26 countries and territories under a single simulated novel pathogen scenario tests command-and-control structures, international data-sharing protocols, and surge capacity — all areas that were found wanting during the COVID-19 pandemic.
Global Health Security
WHO Exercise Polaris II Concludes: WHO's two-day high-level simulation exercise wrapped on April 27, involving 26 countries and territories, 600 health emergency experts, and more than 25 partners. The exercise scenario centered on a fictional novel bacterium to stress-test international outbreak response coordination — from early detection through containment and communications. Findings are expected to feed directly into WHO's preparedness strategy development.
FDA Convenes Advisory Committee on COVID-19 Vaccine Composition: The FDA announced a forthcoming VRBPAC public meeting to deliberate the 2026–2027 COVID-19 vaccine antigen composition for the United States, with a public comment docket now open. This annual process shapes which SARS-CoV-2 variants vaccine manufacturers will target in the upcoming booster cycle.
U.S. Vaccine Advisory Infrastructure in Limbo: A federal court ruling halting the HHS advisory panel under Secretary Kennedy has left COVID and flu vaccine guidance for the upcoming season unclear. Reuters reported that the ruling has created uncertainty for insurers about coverage obligations for newly approved vaccines — a downstream effect with significant implications for vaccine uptake heading into fall 2026.

What to Watch Next
-
VRBPAC COVID-19 Composition Meeting: The upcoming FDA advisory committee session on 2026–2027 COVID-19 vaccine antigen composition will determine which variant strains manufacturers must target. Its outcome will drive booster campaign planning and insurance coverage decisions — especially critical given the current policy vacuum left by the court-halted advisory panel.
-
U.S. Vaccine Coverage Resolution: The legal and policy fallout from the court ruling that froze Kennedy's HHS advisory panel is still unfolding. Watch for court decisions or executive actions that could restore or restructure the federal vaccine recommendation pipeline before the fall 2026 respiratory season begins.
-
Exercise Polaris II Findings Publication: WHO is expected to publish after-action findings and recommendations from the Exercise Polaris II simulation. These findings will likely shape international pandemic preparedness frameworks and could influence funding priorities for health security infrastructure in member states.
This content was collected, curated, and summarized entirely by AI — including how and what to gather. It may contain inaccuracies. Crew does not guarantee the accuracy of any information presented here. Always verify facts on your own before acting on them. Crew assumes no legal liability for any consequences arising from reliance on this content.